How to Get a DME License

Obtaining a DME license is a major achievement for any healthcare provider. But the regulatory process can seem like a complicated maze.
This guide outlines the key steps to obtain your DME certification, stay compliant, and use automation to keep your operations running efficiently.
How to Get a DME License and Why
Entering the medical equipment industry is a fulfilling way to support patient care. It also means navigating a network of legal and professional standards.
A DME license gives you the legal permission to supply life-enhancing equipment to patients. This guide covers the state and federal landscape so your business is built on a solid foundation. If you are just starting your journey, you may also want to check out our deep dive on how to start a DME business.
What is a DME License?
A DME license (Durable Medical Equipment license) is a permit required by state and federal authorities. It allows you to sell, rent, or distribute medical devices intended for long-term use.
This license ensures that DME suppliers meet specific safety, storage, and professional standards. Without it, you can’t legally dispense equipment like oxygen concentrators, wheelchairs, or CPAP machines. You also can’t bill private insurance or government programs for DME.
Which states require a DME license?
The DME license requirements vary a lot by state. Most states require a specific license, while a few are considered "non-licensing" states.
The DME Licensing Landscape: A State-by-State Breakdown
Federal DMEPOS accreditation sets a baseline for Medicare billing. But state-level rules add another layer of complexity. Every state has its own structure. Staying compliant requires understanding whether you need a license to sell to consumers, facilities, or both.
The map below provides a high-level overview of these requirements. It helps identify where you might need specific legal authorization to ship equipment, bill insurance, or operate a physical location.
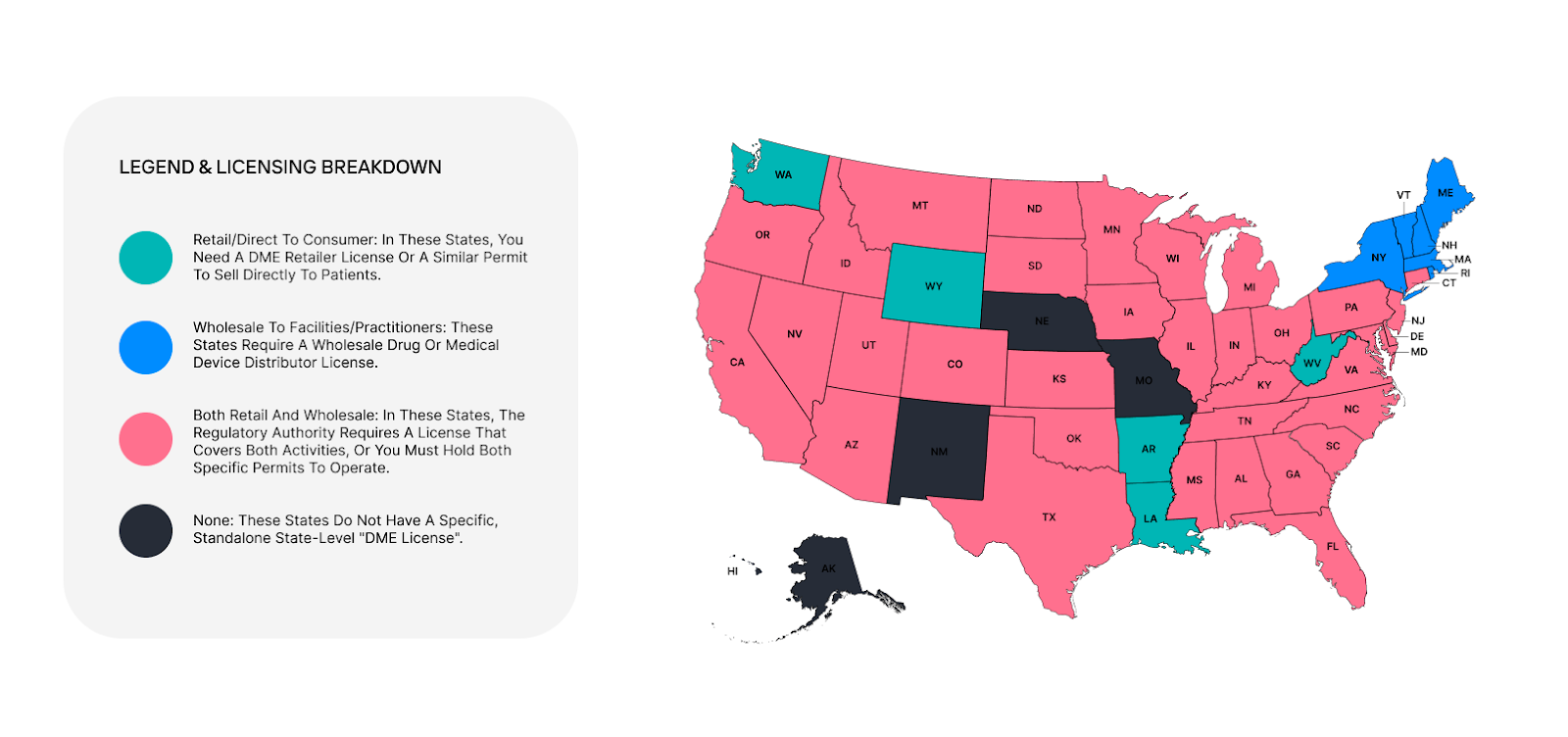
States Without a Standalone "DME License"
Don't be misled by the "no license required" tag. In places such as New York, Nevada, and Arizona, you still encounter tough challenges:
- New York: There is no specific "DME retail license," but you must obtain an NPI and complete Medicaid enrollment (Category of Service 0321) through eMedNY for reimbursement. Additionally, you must meet strict storefront standards to maintain Medicare billing privileges.
- Nevada: While they don't have a "DME License," the Nevada State Board of Pharmacy requires a Medical Products Provider (MDEG) permit for anyone selling or leasing medical devices.
- Arizona: Arizona requires a Medical Gas/DME Permit through the Board of Pharmacy. Even if the state doesn't call it a "license," you cannot ship equipment without this permit.
How to get a DME License in California?
In California, the DME license is managed by the Department of Health Care Services (DHCS) and the Home Medical Device Retailer (HMDR) program. You must submit a PAVE application, provide proof of $300,000 liability insurance, and pass a site inspection. California's DME license requirements often require a "designated representative" to be physically present at your location.
How to get a DME License in Florida?
The DME license process in Florida is overseen by the Agency for Health Care Administration (AHCA). You must undergo a level 2 background screening (fingerprinting) for all owners and managers. Additionally, license applicants must provide a $50,000 surety bond if they are not already Medicare-enrolled.
How to get a DME License in Texas?
To satisfy DME license requirements in Texas, you must register with the Texas Department of State Health Services (DSHS). Texas requires a "Device Distributor" license. If you are a specialized DME provider located in Texas, you must maintain a physical location that is open for inspection during business hours.
How to get a DME License in Georgia?
Georgia requires DME certification through the Georgia Board of Pharmacy. If you provide medical gases (like oxygen), you may need a specialized Medical Gas Dispenser license in addition to your standard retail permit.
How to get a DME License in North Carolina?
The North Carolina Board of Pharmacy issues DME permits. A unique requirement here is the "Person-In-Charge" (PIC) designation. You'll need to name a specific individual responsible for compliance who must submit a passport-quality photo and professional credentials.
How to get a DME License in Pennsylvania?
Pennsylvania requires registration with the Department of Health. While some states focus on the retail aspect, Pennsylvania's oversight is extensive for any DME supplier who maintains inventory within the state borders.
How to get a DME License
The path to becoming a fully licensed DME supplier involves three main pillars: business formation, federal accreditation, and state licensure.
Establish Your Business
First, form a legal entity (LLC or Corp) and obtain an Employer Identification Number (EIN). You must also secure a physical location that meets "Supplier Standards". This means having a 200-square-foot space, a working telephone, and posted hours of operation.
Obtain Federal Certification
To bill Medicare, you must get DMEPOS accreditation from a CMS-approved body (like ACHC or BOC). As of January 1, 2026, CMS now requires annual surveys instead of the previous three-year cycle. You must also obtain a National Provider Identifier (NPI) and post a $50,000 surety bond.
Apply for the State DME License
Once your federal foundation is set, apply to your state board. This usually requires submitting your CMS accreditation proof, insurance certificates, and passing a local site inspection to ensure your HIPAA compliance and storage protocols are in place.
How Long Until Your DME Business is Fully Licensed?
On average, the process takes 4 to 9 months.
- Accreditation: 3 to 5 months
- State licensing: 1 to 2 months
- Medicare enrollment (Form 855S): 60 to 90 days
DME Licensing and Accreditation Fees
Budgeting for the DME license cost is essential for a new business.
Accreditation Complete? Power Your DME with AI Automation
Getting licensed opens the door. Now the real work begins — building operations that maximize revenue and cut administrative burden.
Manual document processing creates bottlenecks. Prior authorization delays hold up revenue. Claim denials drain resources. These operational challenges compound quickly as your business grows.
Notable Systems provides AI-powered automation for DME operations, helping licensed suppliers transform their revenue cycle from day one. Our Intake Manager processes orders in seconds, automatically extracting patient information from any format. Claims Manager evaluates orders against payer-specific coverage criteria before submission, flagging documentation gaps that would trigger denials.
Getting licensed opens the door. Building efficient, AI-powered operations is what drives sustainable growth in the competitive DME landscape.

